A common theory of human evolution says that after our ancestors descended from the trees, they went form walking on four legs to two. But a new study in orang-utans could overturn that theory, by suggesting that our ancestors evolved a bipedal walk while they were still in the trees.
 Walking on two legs, or bipedalism, immediately sets us apart form other apes. It frees our arms for using tools and weapons and is a key part of our evolutionary success. Scientists have put forward a few theories to explain how our upright gait evolved, but the ‘savannah theory’ is by far the most prolific.
Walking on two legs, or bipedalism, immediately sets us apart form other apes. It frees our arms for using tools and weapons and is a key part of our evolutionary success. Scientists have put forward a few theories to explain how our upright gait evolved, but the ‘savannah theory’ is by far the most prolific.
It’s nicely illustrated by this misleading image that has become a mainstay of popular culture. It suggests that our ancestors went from four legs to two via the four-legged knuckle-walking gait of gorillas and chimps. Dwindling forests eventually pushed them from knuckle-walking to a full upright posture. This stance is more efficient over long distances and allowed our ancestors to travel across open savannahs.
But this theory fails in the light of new fossils which push back the first appearance of bipedalism to a time before the forests thinned, and even before our ancestors split from those of chimpanzees. Very early hominins, including Lucy (Australopithecus afarensis) and Millennium Man (Orrorin) certainly ambled along on two legs, but they did so through woodland not plains.
Our arms provide a further clue. Even though our ancestors’ back legs quickly picked up adaptations for bipedalism, they steadfastly kept long, grasping arms, an adaptation more suited to moving through branches. To Susannah Thorpe at the University of Birmingham, these are signs that bipedalism evolved while our ancestors were still living in trees.
Two legs good, four legs bad?
 But there is a snag – an adaptation must provide some sort of benefit. And, as many children painfully discover, it is hard to imagine how walking on two legs could benefit sometime in a tree.
But there is a snag – an adaptation must provide some sort of benefit. And, as many children painfully discover, it is hard to imagine how walking on two legs could benefit sometime in a tree.
But Thorpe has an answer to this too. She spent a year in the Sumatran jungle, studying the orang-utan – the only great ape to spend the majority of its life in the trees.
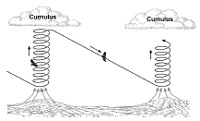
She carefully documented over 3,000 sightings of wild orang-utans moving through the treetops. On large sturdy branches, they walk on all fours (below right), and on medium-sized ones, they start to use their arms to support their weight.
But on the thinnest and most unstable branches, the apes use a posture that Thorpe calls ‘assisted bipedalism’ (below left). They grip multiple branches with their long, prehensile toes and use their arms to balance and transfer their weight. And unlike chimps which bend their knees while standing up, bipedal orang-utans keep their legs straight, just like humans do.

It’s a win-win posture – the hands provide extra safety, while the two-legged stance frees at least one hand to grab food or extra support. With it, the apes can venture onto the furthest and thinnest branches, which provides them with several advantages.
As Thorpe says, “Bipedalism is used to navigate the smallest branches where the tastiest fruits are, and also to reach further to help cross gaps between trees.” That saves them energy because they don’t have to circle around any gaps, and it saves their lives because they don’t have to descend to the ground. “The Sumatran tiger is down there licking its lips”, she said.
A new view of ape & human evolution
With these strong adaptive benefits, it becomes reasonable to suggest that bipedalism evolved among the branches. Based on this theory, Thorpe, along with Roger Holder and Robin Crompton from the University of Liverpool, have painted an intriguing new picture of ape evolution.
It begins in the same way as many others – with the rainforests of the Miocene epoch (24 to 5 million years ago) becoming increasingly patchy. For tree-dwelling apes, the gaps in the canopy started becoming too big to cross. But in Thorpe’s view, these ancestral apes were already using a bipedal stance, and different groups took it in separate directions.
 The ancestors of orang-utans remained in the increasingly fragmented canopy and became specialised and restricted there. The ancestors of chimps and those of gorillas specialised in climbing up and down trees to make use of food both in the canopy and on the ground. The postures used in vertical climbing are actually very similar to those used in four-legged knuckle-walking and this became their walk of choice on the ground.
The ancestors of orang-utans remained in the increasingly fragmented canopy and became specialised and restricted there. The ancestors of chimps and those of gorillas specialised in climbing up and down trees to make use of food both in the canopy and on the ground. The postures used in vertical climbing are actually very similar to those used in four-legged knuckle-walking and this became their walk of choice on the ground.
The ancestors of humans abandoned the trees altogether. They used the bipedal stance that served them well on thin branches to exploit the potential of the stable land environment. Over time, they brought in further adaptations for efficient walking, culminating in the human walking style that we now neglect by sitting at a computer all day.
Thorpe’s reconstruction is delightfully non-human-centric. It suggests that in the evolution of movement, we were conservatives who relied on a walk that had been around for millions of years. Chimps and gorillas with their fancy new knuckle-dragging gait were the true innovators.
Reference: Thorpe, Holder & Crompton. 2007. Origin of human bipedalism as an adaptation for locomotion on flexible branches. Science 316: 1328-1331.
Image: Black and white image from Science magazine.
Technorati Tags: human evolution, apes, orang-utans, bipedalism, walking on two legs,
Related stuff:
On ape and human evolution:
Chimps show that actions spoke louder than words in language evolution
Hidden ‘junk’ gene separates human brains from chimpanzees
Chimps have more adaptive genetic changes than humans
On the evolution of movement:
Salamander robot walks, swims and sheds light on evolutionary step from sea to land
Microraptor – the dinosaur that flew like a biplane
Spread the word: Digg this Del.icio.us Reddit Google Bookmarks Stumbleupon
Filed under: Animal evolution, Animal kingdom, Animal movement, Being human, Chimpanzees, Evolution, Evolutionary arms race, Human evolution, Mammal evolution, Mammals, Primates | 7 Comments »
![]() From an animal’s point of view, the most important things in the world around it are arguably other animals. They provide mates, food, danger and companionship, so as an animal gazes upon its surroundings, it pays for it to be able to accurately discern the movements of other animals. Humans are no exception and new research shows that we are so attuned to biological motion that babies just two days old are drawn to extremely simple abstract animations of walking animals.
From an animal’s point of view, the most important things in the world around it are arguably other animals. They provide mates, food, danger and companionship, so as an animal gazes upon its surroundings, it pays for it to be able to accurately discern the movements of other animals. Humans are no exception and new research shows that we are so attuned to biological motion that babies just two days old are drawn to extremely simple abstract animations of walking animals. Animals move with a restrained fluidity that makes them stand out from inanimate objects. Compared to a speeding train or a falling pencil, animals show far greater flexibility of movement but most are nonetheless constrained by some form of rigid skeleton. That gives our visual system something to latch on to.
Animals move with a restrained fluidity that makes them stand out from inanimate objects. Compared to a speeding train or a falling pencil, animals show far greater flexibility of movement but most are nonetheless constrained by some form of rigid skeleton. That gives our visual system something to latch on to.
















 Like Spider-Man, most spiders can climb sheer surfaces and they do so with two techniques. The most obvious are small claws, called tarsi, that grip onto rough surfaces. Going down in scale, their feet also end in thousands of tiny hairs. These hairs make such close contacts with the microscopic troughs and crests of seemingly smooth surfaces that they stick using the same forces that hold individual molecules together. .
Like Spider-Man, most spiders can climb sheer surfaces and they do so with two techniques. The most obvious are small claws, called tarsi, that grip onto rough surfaces. Going down in scale, their feet also end in thousands of tiny hairs. These hairs make such close contacts with the microscopic troughs and crests of seemingly smooth surfaces that they stick using the same forces that hold individual molecules together. .


